The Research on Modeling and Simulation of TFE Polymerization Process. Key Laboratory of Advanced Control and Optimation for Chemical Processes of Ministry of Education, Shanghai 2. China. 2School of Information Science and Engineering, East China University of Science and Technology, Shanghai 2. China. Received 5 January 2. Revised 1. 0 March 2. Accepted 1. 3 March 2. Published 1. 6 April 2. Copyright . This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. PTFE (polytetrafluoroethylene) is the fluorinated straight- chain polymer, made by the polymerization of tetrafluoroethylene monomer; it is used widely because of its excellent performance and can be obtained by the polymerization of body, solutions, suspensions, and emulsions. But only the last two are the main ways. This research paper makes simulation based on Polymer Plus. It uses the emulsion polymerization method at background to carry out a semibatch reactor system. Upon the actual production conditions, simulation process under the steady state conditions is used to analyze the effects of the changes on operating conditions; the corresponding dynamic model is created to analyze the impact of the changes of conditions on the entire system. Moreover, the amount of APS which plays an important part in this reaction is discussed for getting the most suitable amount of initiator. Because of less research work on this job, it is so difficult to find the related data from the literature. Therefore, this research will have a great significance for the process of the tetrafluoroethylene emulsion polymerization in the future. Past studies, the vapor deposition polymerization (VDP) process was refined empirically and. 5.5 Modeling the Solid-State Reactions of PMDA and ODA.
Introduction. Polytetrafluoroethylene is the most important species in the fluorine- containing polymer and it is applied in many industrial areas, such as aerospace and medical areas. In China, PTFE is mainly obtained from suspension polymerization, but the PTFE from dispersion polymerization . There is a great gap in the performance of the dispersion resin compared with the foreign countries. With the rapid development of industry, the size of the PTFE’s demand has become a national economic indicator, so dealing with the control system of the production process of PTFE not only can improve the quality of the product and reduce energy consumption during the reaction, but also can provide guidance and advise to the scene of the process operation. The PTFE emulsion obtained from TFE dispersion polymerization has been applied in many important areas; foreign researchers have done a lot of work in the synthesis of fluorinated polymer emulsion and made good results in the application of research . Some patents about it are implied in the industrial area successfully. The countries in which science and technology are relatively developed have their own polymerization system. They have a very detailed presentation and research on the performance and application. However, they keep the process conditions and the recipe confidential; only a few patents involved are about this; most of the articles are the analysis of the synthesized product. Therefore, this paper about dispersion polymerization of TFE has a practical significance for the industrial craft process in domestic. 
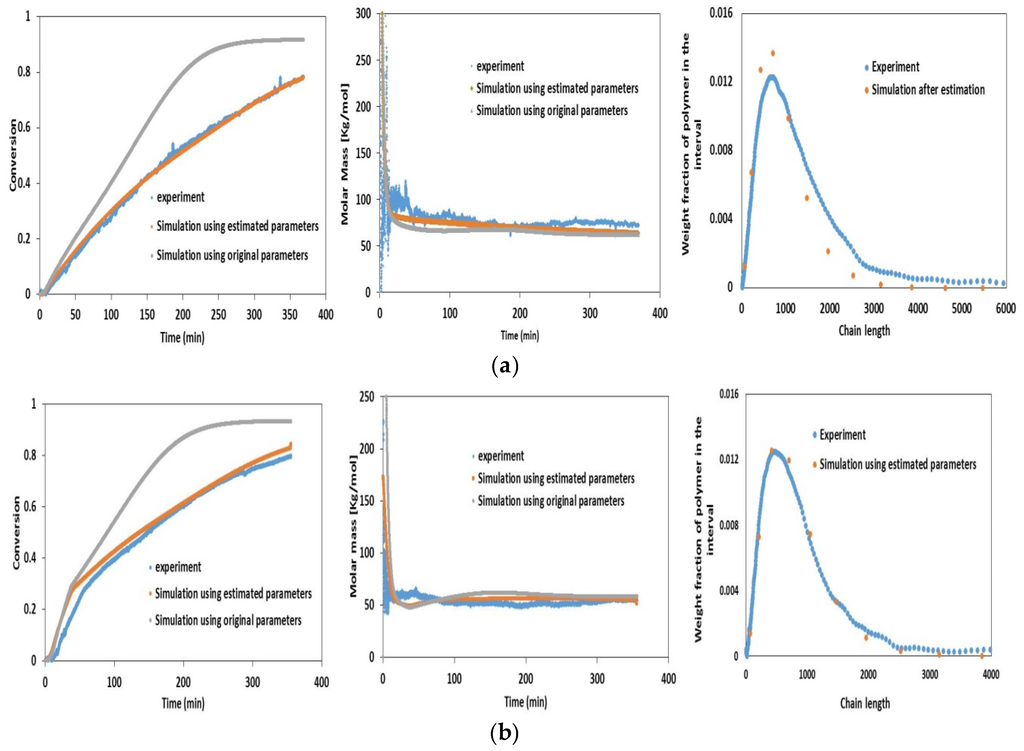
PDF polymerization process modeling methods in stereochemical analysis by neil a dotson 1996-01-01 PDF modeling & simulation of polymerization reactionprocess reactor.Process Background of TFE Dispersion Polymerization. The main object of this study is the polymerization process of tetrafluoroethylene which is commonly used in industry. It mainly uses the emulsion polymerization and semicontinuous feeding methods . Because the initiator was decomposed under the acid condition, we add some glacial acetic acid to balance the liquid acid. Besides, we also should add stabilizer paraffin and chain transfer agent DDM; the main process can be summarized as follows: adding the deionized water and accessory ingredient (including initiators, stabilizers, modifiers, and chain transfer agent) which compounded well in advance (see Section 3. After that we stop stirring and cooling the reactor. During the total process, the temperature should be kept between 8. The static simulation process is shown in Figure 1. Figure 1: Process of the polymerization. Steady- State Process Simulation and Analysis. Polymerization Reaction Components. The chemical substances were divided into conventional, segment, oligomer, and polymer in polymer plus. They have the properties data like the boiling and melting point, the molecular structure, and molecular weight. Segment mainly includes repetitive unit, endpoint, and grafting site. The properties of the polymer are mainly dependent on the polymer segment type, quantity, and composition forms. The reaction components in this paper are shown in Table 1. Table 1: The components involved in the reaction. In this process, APS is 5 g, CH3. COOH is 2. 50 m. L, DDM is 1. And the feed temperature of TFE is 3. Polymerization reactor is the main place of reaction, so its parameter setting is the most critical and important; in this paper, volume is set to 1. Since this reaction is a semicontinuous process, we make the reaction stop when PTFE yields reach to 1. Polymerization Kinetics. The polymerization of tetrafluoroethylene is radical polymerization . The kinetic equation is expressed as follows: (1)chain initiator: ; (2)chain growth: ; (3)chain transfer: transfer to monomer: ; transfer to the regulator: ; transfer to chain transfer agent: ; (4)chain termination: ; The first step includes two parts: initiator decomposing to radical ion, combining with monomer to be monomer radical. The second step is the process of chain transfer and the third step is the process of transferring to monomer, chain transfer agent, and regulator. The end step is the process of coupling termination. This paper combines the relative literature data and the correction in the actual simulation we have done, so it gets a better simulation effect. The polymerization of TFE is free radical polymerization, although Polymer Plus provides relevant radical polymerization model, but the kinetics parameters of different reactions are different and because of the diversity of PTFE process; kinetics parameters debugging is very difficult and time consuming. This paper combines literature data and the results of the simulation based on the actual correction data; therefore, it gets a better simulation results. Table 2 is the setting of the kinetic parameters. Table 2: Reaction rate and preexponential factor setting. The Analysis of the Steady State. The Effect of Reactor Temperature on the Product. For the polymerization reaction, the reaction temperature directly affects the polymerization degree which reflects the quality of the product. Too low temperature will lead to slow response and the product with low intensity. On the contrary, the reaction rate will be rapid and the reaction is difficult to control. Low temperature will get low monomer conversion rate, because low molecular motion is not conducive to the proliferation of free radicals and the collision between monomers and radical. With the rises of the temperature, the chain growth rate constant and the number of the growth active centers both increase and the probability of spreading and collision between monomer and active radicals are elevated; meanwhile, the amount of PTFE increases leads to conversion rate of the monomer increasing. However, when the temperature reaches a certain stage, the curve of conversion rate will gradually tend to level because of the secondary reaction increasing, such as coupling termination and disproportionation termination. With the reaction proceeds continuing, the concentration of active centers is reduced, so the reaction rate is leveled off. The size of the molecular weight reaches unanimity basically as time increases. In order to investigate the impact of temperature on the molecular weight distribution of the polymer, the reactor temperature is set to 8. It is obvious that the increase of the temperature leads to the decrease of the molecular weight of polymer. The Effect of the Initiator on the Product. Figure 5 shows the PTFE dispersion products molecular weight when APS is between 2 g and 8 g. Initiator is one of the most important part of emulsion formulations; it cannot be too much or too little. The reaction speed will be too fast if its usage is too much, on the contrary, too little will make it difficult to trigger the reaction and is not good for the polymer performance. Figure 5: Effects of initiator flow on PDI. Product conversion rate will increase gradually when initiator increases, and the rise of the primary free radicals will increase the frequency of chain initiator and then lead to the rise of the monomers conversion as well as reaction rate. Meanwhile, in the chain termination and chain transfer stage, the rise of the active end of the initiation phase accelerates the collision probability between primary radicals and active chain. Therefore, the length of the molecular chain becomes short; it means that PDI reduces gradually. However, too much initiator will be treated as the electrolyte and reduce the stability of the emulsion polymerization process. The Effect of the Regulator Feed on the Product. Since the initiator agent decomposed under the acidic conditions, its half- life will be shortened when PH value reduces. Hence we add the right amount of glacial acetic acid in the reactor to adjust the acidity of the liquid. Figure 6 shows that PDI increases and the monomer conversion reduces as CH3. COOH increases; this is because CH3. COOH has a great effect on the polymerization reaction, especially in the liquid phase; it will be the chain transfer agent in the chain- transfer reaction. It is easy to react with growing macromolecular radicals and terminate the active chain, and the decrease of chain growth monomer will lead to the conversion rate reducing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2016
Categories |
 RSS Feed
RSS Feed
